Relevant information for your patients
This section gives you some ideas to inform your patients clearly and comprehensively about why undergo a prenatal test to detect main fetal chromosomal aneuploidies. For patients it can actually be difficult to choose which tests to rely on, in addition to the compulsory pregnancy tests, so it is helpful for you to be properly prepared to answer all their questions. Sorgente Genetica has collected and summarized for you the most common questions from pregnant women and the related answers.
Why perform AURORA test?
AURORA is a non-invasive prenatal test, completely safe for both mother and baby. This test allows detecting the main chromosomal abnormalities that may be present in the fetus, with reliability higher than 99.9% for the most common trisomies (i.e. trisomy 21 and trisomy 18). For other trisomies, click here.
Who can undergo the Aurora test and when is it performed?
All expectant mothers can undergo this test starting from the 10th week of pregnancy. The test can be performed for singleton or twin pregnancies (either monozygotic or dizygotic) and on women of all ages and ethnic group.
What does the Aurora test detect?
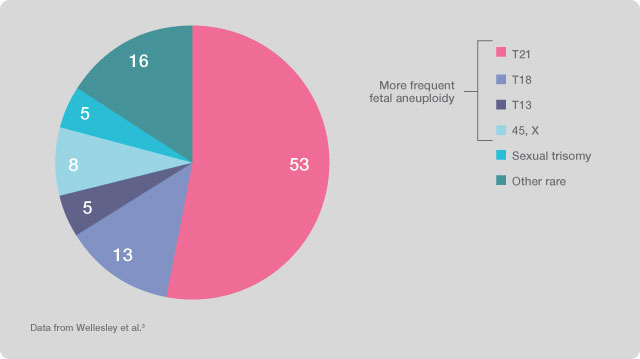
The test only evaluates the presence of chromosomal aneuploidies related to chromosomes 21 (Down Syndrome, three copies of chromosome 21, with an incidence of 1/700 live births), 18 (Edwards Syndrome, three copies of chromosome 18, with an incidence of approximately 1/7,000 live births), 13 (Patau Syndrome, three copies of chromosome 13, with an incidence of 1/10,000 live births), aneuploidies of sexual chromosomes (X chromosome monosomy or Turner Syndrome, Trisomy X, XXX, Klinefelter Syndrome, XXY, and Jacobs Syndrome, XYY) and, at the specific request of the patient, the aneuploidies of chromosomes 9 and 16 can also be added, as well as the main microdeletions (loss of a chromosome region), in particular Di George Syndrome (deletion 22q11), Angelman/Prader-Willi Syndrome (deletion 15q11), deletion 1p36, Wolf-Hirschhorn Syndrome (deletion 4p) and Cri-du-chat Syndrome (deletion 5p). At the discretion of the patient, the test can also determine the baby’s sex.
How does the Aurora screening test work?
The AURORA test is performed on a blood sample of the expectant mother because, during pregnancy, there are DNA fragments of fetal origin circulating in the maternal blood, which are detectable starting from the 5th week of pregnancy and whose concentration increases throughout pregnancy. The fetal DNA is then degraded after childbirth. In order to achieve an amount of fetal DNA sufficient to ensure high specificity and sensitivity of the test we just need to wait until the 10th week of pregnancy. The treatment of the collected sample allows us to analyze the plasma component of maternal blood containing cell free fetal DNA.
How reliable is the AURORA test?
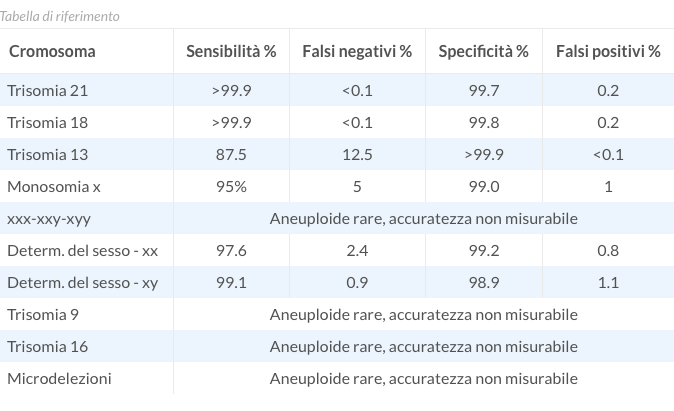
The AURORA non-invasive prenatal test has shown 99% specificity for all main fetal aneuploidies, with over 99.9% reliability in detecting the most common aneuploidies, i.e. Trisomy 21 and Trisomy 18. For Trisomy 13 and Monosomy X too, sensitivity rates are high (87.5% and 95%, respectively). In addition, for the most frequent trisomies the rate of false negatives and false positives is less than 0.1% and 0.3%, respectively.
Does the Aurora test replace the first and second quarter screening test?
The AURORA prenatal test is much more reliable than traditional first and second quarter tests (double test, triple test, etc.); however, it does not replace invasive prenatal diagnostic tests (amniocentesis and chorionic villous sampling), which should be performed in case of successful result of the AURORA test.
Why choose AURORA instead of other tests?
The AURORA test uses an advanced technology that allows the massive parallel sequencing (MPS) of the entire genome. To perform this test we use the sequencer NGS (Next Generation Sequencing) Illumina HiSeq 2500, a leading quality system in DNA sequencing. Thanks to this technology, specific chromosomal regions are sequenced at high processivity (about 30 million sequences). The sequences are then quantified via an advanced bioinformatics analysis, according to the SAFER ™ VerinataHealth algorithm, which is able to detect the presence of possible fetal aneuploidies.
Therefore, the use of this technology allows:
- maximum reliability of results (>99.9% for the most widespread trisomies, i.e. Trisomy 21 and 18);
- reduction of false positives (<0.3% for the most widespread trisomies, i.e. Trisomy 21 and 18);
- reduction to a minimum of null tests (<0.07%, i.e. less than 1 in 1,000 patients).
We also guarantee:
- report delivery in approximately 10 days;
- clear test results;
- reports drawn up by qualified geneticists.
What does the AURORA service include?
The customer can choose between the two following options:
- AURORA test:
- trisomy 21, 18 and 13;
- main aneuploidies of sexual chromosomes (X chromosome monosomy or Turner Syndrome, Trisomy X, Klinefelter Syndrome, XXY, and Jacobs Syndrome, XYY);
- AURORA test + :
- trisomy 21, 18, 13 and the main aneuploidies of sexual chromosomes
- aneuploidies of chromosomes 9 and 16;
- main microdeletions (loss of a chromosome region), in particular: deletion 22q11 (Di George Syndrome), deletion 15q11-q13 (Angelman/Prader-Willi Syndrome), deletion 1p36, deletion 4p (Wolf-Hirschhorn Syndrome) and deletion 5p (Cri-du-chat Syndrome);
The family can decide whether or not to know the sex of the fetus for both options mentioned above.
The service also includes genetic counselling before and after testing. Doctors and geneticists, making use of the informed consent form, illustrate to patients what the test is used for, what results can be achieved and how to interpret the test results.
What are fetal aneuploidies?
Aneuploidies are chromosomal abnormalities characterized by an alteration in the number of chromosomes compared to the normal human female karyotype 46, XX and the male karyotype 46, XY. In fact, what we define by trisomy is the presence of a an extra chromosome and by monosomy the absence of a chromosome.
Trisomy 21 or Down syndrome (karyotype 47, XX or XY, +21), is caused by the presence of a supernumerary chromosome 21 compared to the normal chromosomal structure. This abnormality occurs in 1 in every 700 newborns. Down syndrome is associated with heart defects, delay in cognitive skills and growth; from a clinical point of view, the events associated with the syndrome are heterogeneous and unpredictable before birth.
Trisomy 18 or Edwards syndrome (karyotype 47, XX or XY, +18), is caused by the presence of a supernumerary chromosome 18 compared to the normal chromosomal structure. This abnormality occurs in 1 out every 7,000 newborns. It is characterized by severe mental retardation, as well as congenital heart and kidney defects and other pathological conditions. The life expectancy of a newborn suffering from Trisomy 18 is particularly low, rarely exceeding the first year of age.
Trisomy 13 or Patau syndrome (karyotype 47, XX or XY, +13) is caused by the presence of a supernumerary chromosome 13 compared to the normal chromosomal structure. This abnormality affects 1 in every 10,000 new-born babies. It is characterized by severe psychomotor retardation, heart defects, and other conditions. Life expectancy is similar to that observed for trisomy 18.
Monosomy X (45, X) or Turner syndrome. It is the only monosomy compatible with life; the individual is phenotypically female and has only one X chromosome instead of two. This aneuploidy involves short stature, delayed or absent puberty, infertility, heart or kidney problems and possible learning difficulties.
Karyotype 47, XXY or Klinefelter syndrome; the individual is phenotypically male, with two X chromosomes and one Y chromosome. This syndrome involves stature higher than average, delayed puberty, infertility and, in some cases, learning difficulties.
Karyotypes 47, XXX (Triple X) and 47, XYY (Jacobs Syndrome). Are not associated with obvious defects at birth and can often remain undiagnosed.
What are microdeletions and microduplications?
By the term microdeletion and microduplication we refer to chromosomal abnormalities characterized by loss or duplication of a small part of a chromosome. Syndromes caused by these abnormalities are characterized by phenotypic abnormalities of varying extent, depending on the chromosome involved, on the affected region and on the scope of the lost or duplicated region.
AURORA provides you with informational material that you can share with your patients to help them in their choice:
- AURORA information brochure;
- Informational video on the importance of non-invasive prenatal tests, click here.
This section gives you some ideas to inform your patients clearly and comprehensively about why undergo a prenatal test to detect main fetal chromosomal aneuploidies. For patients it can actually be difficult to choose which tests to rely on, in addition to the compulsory pregnancy tests, so it is helpful for you to be properly prepared to answer all their questions. Sorgente Genetica has collected and summarized for you the most common questions from pregnant women and the related answers.
Why perform the AURORA test?
AURORA is a non-invasive prenatal test, completely safe for both mother and baby. This test allows detecting the main chromosomal abnormalities that may be present in the fetus, with reliability higher than 99.9% for the most common trisomies (i.e. trisomy 21 and trisomy 18). For other trisomies,click here.
Who can undergo the AURORA test and when is it performed?
All expectant mothers can undergo this test starting from the 10th week of pregnancy. The test can be performed for singleton or twin pregnancies (either monozygotic or dizygotic) and on women of all ages and ethnic group.
What does the AURORA test detect?
The test only evaluates the presence of chromosomal aneuploidies related to chromosomes 21 (Down Syndrome, three copies of chromosome 21, with an incidence of 1/700 live births), 18 (Edwards Syndrome, three copies of chromosome 18, with an incidence of approximately 1/7,000 live births), 13 (Patau Syndrome, three copies of chromosome 13, with an incidence of 1/10,000 live births), aneuploidies of sexual chromosomes (X chromosome monosomy or Turner Syndrome, Trisomy X, XXX, Klinefelter Syndrome, XXY, and Jacobs Syndrome, XYY) and, at the specific request of the patient, the aneuploidies of chromosomes 9 and 16 can also be added, as well as the main microdeletions (loss of a chromosome region), in particular Di George Syndrome (deletion 22q11), Angelman/Prader-Willi Syndrome (deletion 15q11), deletion 1p36, Wolf-Hirschhorn Syndrome (deletion 4p) and Cri-du-chat Syndrome (deletion 5p). At the discretion of the patient, the test can also determine the baby’s sex.
How does the AURORA screening test work?
The AURORA test is performed on a blood sample of the expectant mother because, during pregnancy, there are DNA fragments of fetal origin circulating in the maternal blood, which are detectable starting from the 5th week of pregnancy and whose concentration increases throughout pregnancy. The fetal DNA is then degraded after childbirth. In order to achieve an amount of fetal DNA sufficient to ensure high specificity and sensitivity of the test we just need to wait until the 10th week of pregnancy. The treatment of the collected sample allows us to analyze the plasma component of maternal blood containing cell free fetal DNA.
How reliable is the AURORA test?
The AURORA non-invasive prenatal test has shown 99% specificity for all main fetal aneuploidies, with over 99.9% reliability in detecting the most common aneuploidies, i.e. Trisomy 21 and Trisomy 18. For Trisomy 13 and Monosomy X too, sensitivity rates are high (87.5% and 95%, respectively). In addition, for the most frequent trisomies the rate of false negatives and false positives is less than 0.1% and 0.3%, respectively.
Does the AURORA test replace the first and second quarter screening tests?
The AURORA prenatal test is much more reliable than traditional first and second quarter tests (double test, triple test, etc.); however, it does not replace invasive prenatal diagnostic tests (amniocentesis and chorionic villous sampling), which should be performed in case of successful result of the AURORA test.
Why choose AURORA instead of other tests?
The AURORA test uses an advanced technology that allows the massive parallel sequencing (MPS) of the entire genome. To perform this test we use the sequencer NGS (Next Generation Sequencing) Illumina HiSeq 2500, a leading quality system in DNA sequencing. Thanks to this technology, specific chromosomal regions are sequenced at high processivity (about 30 million sequences). The sequences are then quantified via an advanced bioinformatics analysis, according to the SAFER ™ VerinataHealth algorithm, which is able to detect the presence of possible fetal aneuploidies.
Therefore, the use of this technology allows:
· Maximum reliability of results (>99.9% for the most widespread trisomies, i.e. Trisomy 21 and 18);
· Reduction of false positives (<0.3% for the most widespread trisomies, i.e. Trisomy 21 and 18);
· Reduction to a minimum of null tests (<0.07%, i.e. less than 1 in 1,000 patients).
We also guarantee:
· report delivery in approximately 10 days;
· clear test results;
· Reports drawn up by qualified geneticists.
What does the AURORA service include?
The customer can choose between the two following options:
1. AURORA test:
o trisomy 21, 18 and 13;
o main aneuploidies of sexual chromosomes (X chromosome monosomy or Turner Syndrome, Trisomy X, Klinefelter Syndrome, XXY, and Jacobs Syndrome, XYY);
2. AURORA test + :
o Trisomy 21, 18, 13 and the main aneuploidies of sexual chromosomes
o Aneuploidies of chromosomes 9 and 16;
o Main microdeletions (loss of a chromosome region), in particular: deletion 22q11 (Di George Syndrome), deletion 15q11-q13 (Angelman/Prader-Willi Syndrome), deletion 1p36, deletion 4p (Wolf-Hirschhorn Syndrome) and deletion 5p (Cri-du-chat Syndrome);
The family can decide whether or not to know the sex of the fetus for both options mentioned above.
The service also includes genetic counselling before and after testing. Doctors and geneticists, making use of the informed consent form, illustrate to patients what the test is used for, what results can be achieved and how to interpret the test results.
What are fetal aneuploidies?
Aneuploidies are chromosomal abnormalities characterized by an alteration in the number of chromosomes compared to the normal human female karyotype 46, XX and the male karyotype 46, XY. In fact, what we define by trisomy is the presence of a an extra chromosome and by monosomy the absence of a chromosome.
Trisomy 21 or Down syndrome (karyotype 47, XX or XY, +21), is caused by the presence of a supernumerary chromosome 21 compared to the normal chromosomal structure. This abnormality occurs in 1 in every 700 newborns. Down syndrome is associated with heart defects, delay in cognitive skills and growth; from a clinical point of view, the events associated with the syndrome are heterogeneous and unpredictable before birth.
Trisomy 18 or Edwards syndrome (karyotype 47, XX or XY, +18), is caused by the presence of a supernumerary chromosome 18 compared to the normal chromosomal structure. This abnormality occurs in 1 out every 7,000 newborns. It is characterized by severe mental retardation, as well as congenital heart and kidney defects and other pathological conditions. The life expectancy of a newborn suffering from Trisomy 18 is particularly low, rarely exceeding the first year of age.
Trisomy 13 or Patau syndrome (karyotype 47, XX or XY, +13) is caused by the presence of a supernumerary chromosome 13 compared to the normal chromosomal structure. This abnormality affects 1 in every 10,000 new-born babies. It is characterized by severe psychomotor retardation, heart defects, and other conditions. Life expectancy is similar to that observed for trisomy 18.
Monosomy X (45, X) or Turner syndrome. It is the only monosomy compatible with life; the individual is phenotypically female and has only one X chromosome instead of two. This aneuploidy involves short stature, delayed or absent puberty, infertility, heart or kidney problems and possible learning difficulties.
Karyotype 47, XXY or Klinefelter syndrome; the individual is phenotypically male, with two X chromosomes and one Y chromosome. This syndrome involves stature higher than average, delayed puberty, infertility and, in some cases, learning difficulties.
Karyotypes 47, XXX (Triple X) and 47, XYY (Jacobs Syndrome). Are not associated with obvious defects at birth and can often remain undiagnosed.
What are microdeletions and microduplications?
By the term microdeletion and microduplication we refer to chromosomal abnormalities characterized by loss or duplication of a small part of a chromosome. Syndromes caused by these abnormalities are characterized by phenotypic abnormalities of varying extent, depending on the chromosome involved, on the affected region and on the scope of the lost or duplicated region.
AURORA provides you with informational material that you can share with your patients to help them in their choice:
· AURORA test presentation video,click here;
· AURORA information brochure;
· Informational video on the importance of non-invasive prenatal tests,click here.